
WHO Report on Antibiotic Pipeline and Online Consultation on Monitoring the Global Action Plan
The World Health Organization (WHO) has been visibly busy this month in the area of antimicrobial resistance (AMR). The agency released a report on the global pipeline of new antibiotics (including anti-tuberculosis drugs) on 19th September. It can be downloaded here. There is also a new infographic on what WHO considers priority pathogens in addition to tuberculosis. There is probably little argument about the list as well – Gram-negative bacterial resistance is at the top of the list, which reflects the reality on the ground in most countries in the world including Singapore.

Screenshot of the WHO Priority Pathogen List Infographic
The report has a generally pessimistic slant, estimating that there could be around 10 new antibiotics launched over the next five years, with the caveat that none of these would belong to truly novel classes of antibiotics and expand on the armamentarium against the antibiotic-resistant bacteria, especially those on the critical priority list. Of the current 10 antibiotics with Gram-negative activity in Phase 1 trials now, the report estimated that only 1 or 2 would reach the market in the next decade, based on historical success rates of 14%.
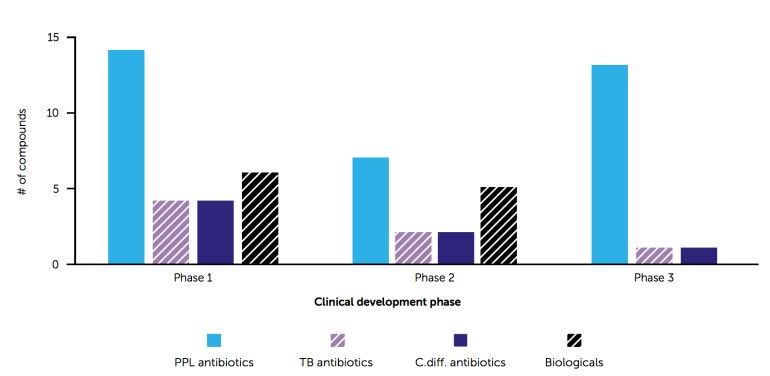
Antibiotic pipeline according to the WHO September 2017 report.
There is another nice infographic representation at the WHO Global Observatory on Health R&D which I have taken the liberty to cut and paste below. It shows clearly the breakdown of the new antibiotics – only 4 and 10 of those drugs in phases I-III of clinical trials are active against Acinetobacter baumannii and Enterobacteriaceae, for example, and few of these are from novel classes of antibiotics.

Antibiotics in the pipeline – screenshot from the WHO Global Observatory on Health R&D.
Of the 4 drugs with known activity against A. baumannii (which is by far the most drug-resistant bacteria in Singapore and other regional hospitals, but which thankfully does not cause overwhelming numbers of infections), 1 is a tetracycline-like antibiotic (TP-6076 from Tetraphase), 2 are siderophore-cephalosporin combinations (Ceferidocol and GSK-3342830 from Shionogi and GlaxoSmithKline – both antibiotics appear to have originated from a collaboration between the two companies in 2010, which has since ended), while the last is a double beta-lactamase-inhibitor (ETX2514-sulbactam). There are 4 other drugs with possible activity against A. baumannii, including another tetracycline and a polymyxin-combination drug, but less data are available at present.
Overall, the report is useful, and offers perhaps more grounds for optimism than it seems to imply. It also announced that there will be a separate report on antibiotic stewardship – WHO Global Framework for Development and Stewardship to Combat Antimicrobial Resistance – that is forthcoming.
The WHO has also put up an online consultation on its “Tripartite Monitoring and Evaluation (M&E) approach” to its Global Action Plan on AMR, with a dateline of 30th September 2017. The actual form is here, but it is only open to institutions, specifically “country governments and organisations with a technical or managerial remit related to national and international responses to AMR, including private sector, industry bodies, research institutions and civil society”. The form is indeed detailed and exhaustive, with many technical aspects. It is indeed important to monitor and evaluate the outcomes of the Global Action Plan on AMR, and the online consultation will be useful as many of the outcome indicators are either difficult to collate – especially for resource-strapped countries – or potentially not useful for assessing the control of AMR.
