
Antibiotic Prophylaxis Against Infective Endocarditis
I was invited to give this talk at the ECHO Singapore 2018 conference last Saturday, and had to do a bit of reading up.
Who should receive antibiotic prophylaxis against endocarditis, particularly when undergoing dental procedures, is obviously an important clinical decision. Endocarditis – inflammation of the inner layer of the heart, particularly the heart valves – is a rare but devastating condition which was previously most commonly caused by viridans streptococci which are found in the oral cavity and gastrointestinal tract (it has been replaced in the past decade or so by Staphylococcus spp.). The rationale for pre-2007 strategies for preventing endocarditis has been summarised by a Canadian review as follows:
- Infective endocarditis is rare but life-threatening, hence prevention is better than treatment.
- Specific, known cardiac conditions predispose to infective endocarditis.
- Bacteraemia with endocarditis-causing organisms can occur in association with invasive dental, gastrointestinal or genitourinary procedures.
- Antimicrobial prophylaxis is likely to be effective in patients who undergo the above procedures.
The main bulk of evidence for such an approach were from old animal model studies (mice and rabbits) in the 1980’s or before.
In the 2000’s, there was more nuanced understanding. Firstly, activities of daily living may result in bacteraemia with endocarditis-causing organisms. In a study published in Circulation in 2008, researchers from the USA showed that bacteraemia could occur in 23% of subjects brushing their teeth, and 33% and 60% of subjects who underwent tooth extraction with amoxicillin or placebo prophylaxis respectively.
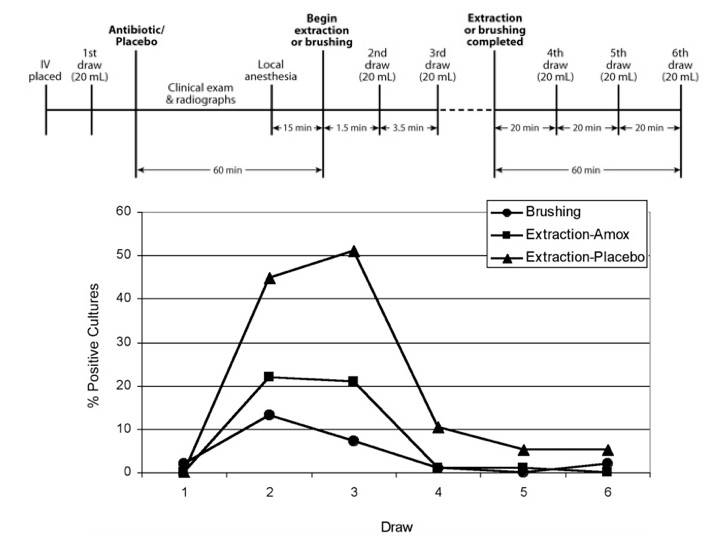
Screen-capture from Dr Lockhart and co-workers’ study published in 2008.
Secondly, other than the concerns of antimicrobial resistance with widespread use of antibiotics, experts factored in the risk that antibiotics could also result in adverse events. Several publications in the USA have shown that antibiotics constitute one of the most common causes for people presenting to the emergency department for drug-related adverse events.

Starting with the 2007 American Heart Association (AHA) guidelines on the prevention of infective endocarditis, attempts were made to stratify cardiac conditions and the accompanying invasive procedures with the highest risk of endocarditis, and to recommend antibiotic prophylaxis for these combinations alone. The approach of the 2015 European Society of Cardiology guidelines was just slightly more conservative than that of the AHA guidelines, but UK’s National Institute for Health and Care Excellence (NICE) took it to an extreme, essentially not recommending antibiotic prophylaxis per se (the word “routinely” was added in the 2016 update to the original 2008 guidelines).

Comparison of major infective endocarditis prophylaxis guidelines. Adapted from Current Infectious Diseases Report 2017;19:9.
The trouble with guidelines on antibiotic prophylaxis for endocarditis is that there is no high-level evidence available – clinical trials are probably impossible to run because of the large sample sizes required.
However, investigators have tried to determine the impact of the guidelines and these have been decidedly mixed, with the majority demonstrating that antibiotic use plummeted, while infective endocarditis rates generally remained stable. However, the majority of the long-term follow-up has been short, and there were two notable exceptions to the results. The first was from Holland, where investigators showed a rise in rates of infective endocarditis since 2007 (after the AHA guidelines were published), although it was not quite satisfactory, since there was no corresponding data on antibiotic use.

Compiled screen captures from the Dutch study tracking endocarditis rates in Holland.
However, the longitudinal data from the UK was more damning, as can be seen from the images below. Endocarditis rates increased dramatically – particularly in “high-risk” patients – after the NICE guidelines were introduced. This corresponded with a sharp decrease in community prescription of antibiotics (article behind a paywall).


Screen capture from the 2015 Lancet article by Dayer, et al.
In summary, antibiotic prophylaxis retains a role in the prevention of infective endocarditis at this stage, although the evidence is mixed, and the main benefit is likely in patients at high-risk of endocarditis. It is crucial to remember that antibiotics do cause side effects on occasion (even single-dose amoxicillin or clindamycin), and overuse will promote antimicrobial resistance.