
Subscription Model for Antibiotics (UK)
A belated post on the announcement on 9th July that UK will be testing a subscription model for antibiotics that hopes to encourage the pharmaceutical industry to develop new antibiotics in the future.

The background to this is that – unlike most other drugs and medical devices – there is acknowledged market failure where antibiotics and antibiotic development are concerned. Prescription of antibiotics usually results in individual good (i.e. bacterial infections are effectively treated in the sick patient) but generates medium- and long-term negative externality in the form of antimicrobial resistance that progressively reduce the effectiveness of antibiotics over time. The response in most parts of the world has been to clamp down (or “police”, as in the case with hospital antimicrobial stewardship) on their use – especially broad-spectrum antibiotics. In an industry where returns are based on volume of sales and drug prices, such restrictions in prescription, coupled with the relatively low prices antibiotics can command (relative to cancer drugs or those that cure rare diseases) and the generally short duration of prescription, has resulted in significant disincentives for the pharmaceutical industry to develop new antibiotics and bring these to market.
There are now a significant number of public and public-private initiatives that aim to spur the development of new (and new classes of ) antibiotics, utilising both “push” (i.e. providing funds) and “pull” (i.e. assurance of a market) incentives. Many of these are nicely listed in the AMR Conference website (screenshot below). The UK initiative will fall into the latter group of incentives, assuring pharmaceutical companies that there will be confirmed revenue regardless of the future volume of sales of these drugs by paying for them upfront.
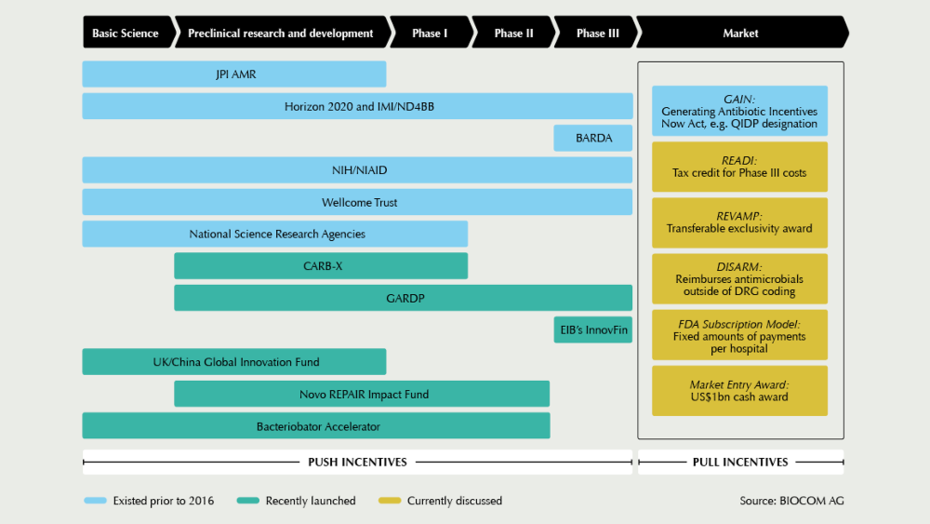
I could not find many details of how the UK NHS and NICE’s experimental subscription model is supposed to work. However, versions of this has already been proposed by others including the FDA (for hospitals in the United States). It is likely that a sufficiently large number of countries will have to implement this model (which works for enterprise software or Netflix, for example) for the pharmaceutical industry to be “incentivised”, however. Singapore – small as the market is – should consider doing the same.
Here is a paper that describes a couple of other models for funding new antibiotics, for those that are interested.